 United States patent 5721067 titled ‘Rechargeable lithium battery having improved reversible capacity’ represents a significant breakthrough by Electrovaya for dealing with the difficult problem of first charge capacity loss with lithium ion cells using carbon as the active anode material. The irreversible losses of carbon have long plagued the lithium ion battery industry and continue to do so to this day for manufacturers not authorized to use Electrovaya’s exclusive breakthrough technology.
United States patent 5721067 titled ‘Rechargeable lithium battery having improved reversible capacity’ represents a significant breakthrough by Electrovaya for dealing with the difficult problem of first charge capacity loss with lithium ion cells using carbon as the active anode material. The irreversible losses of carbon have long plagued the lithium ion battery industry and continue to do so to this day for manufacturers not authorized to use Electrovaya’s exclusive breakthrough technology.
In a traditional lithium ion cell the only source of lithium within the cell upon construction is that which is contained in the cathode material itself such as lithium-cobalt-oxide, lithium-manganese-oxide or lithium-iron-phosphate. These cathode materials are produced in conjunction with lithium containing source ingredients such as lithium carbonate in order to synthesize fully lithiated cathode materials in the discharged state.
The well known and predominant industry challenge of using carbon anode materials is that they irreversibly consume approximately 20% of the lithium received during first charge. Lithium, electrolyte and other additives within the cell are adsorbed during formation of the solid electrolyte interphase layer (SEI) and are locked away in non-active carbon sites reducing the total reversible lithium content of the cell, effecting an immediate and unavoidable increase in mass of under-utilized cathode material, adding to the cell’s cost, size and weight.
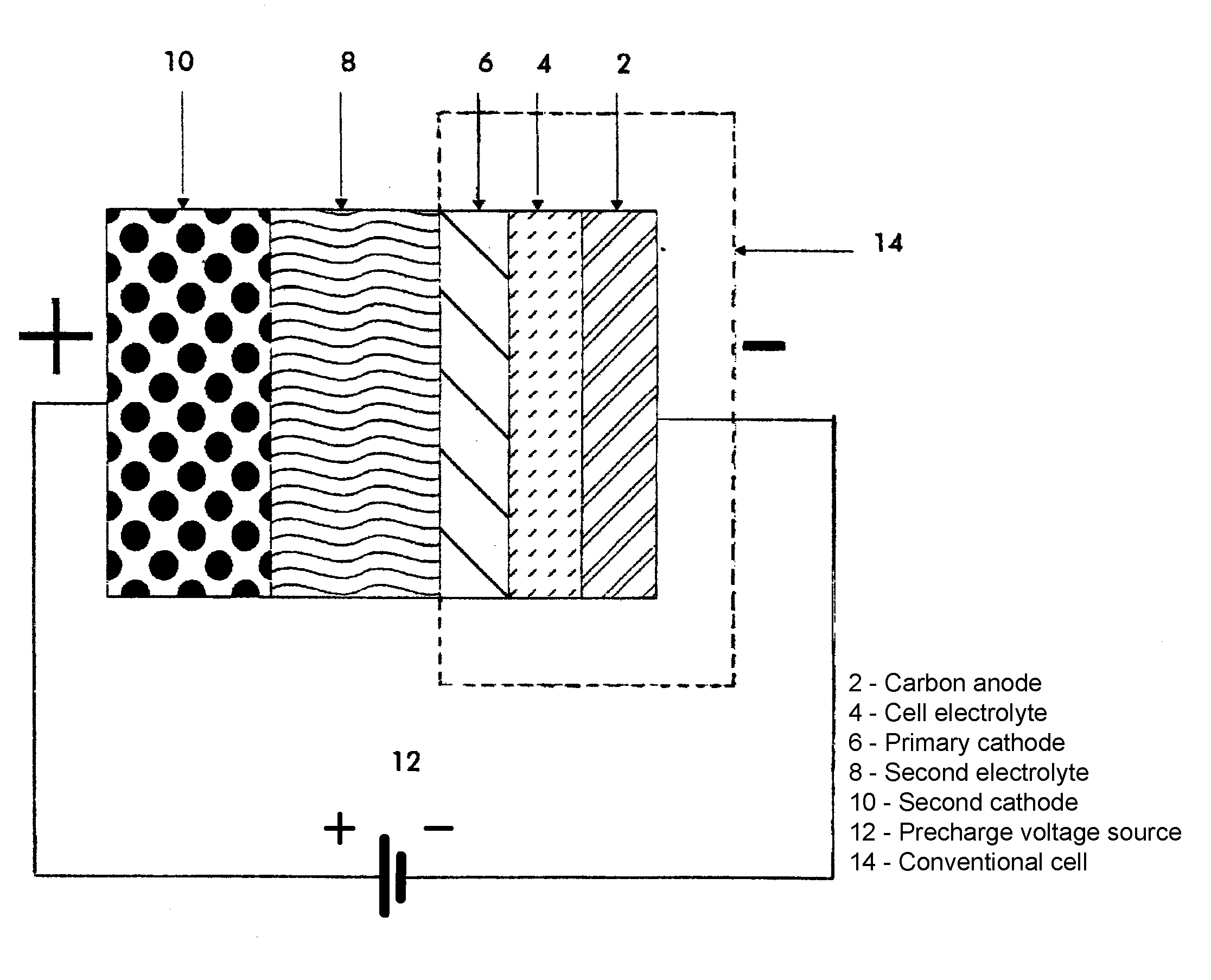
Electrovaya’s unique invention allows these irreversible losses to occur with little to no concern for the amount of lithium and electrolyte consumed during the formation process. By adding a novel twist to their manufacturing technique, Electrovaya has placed a significant portion of their formation process ahead of the final assembly stage. The exact nature of their technology is explained in great detail in their patent, but the main focus of the invention lies in the addition of an extra step in the cell’s manufacturing process whereby assembly of the complete cell is performed as usual, minus the aluminum foil current-collector-laminate normally covering the outer cathode surface.
With the outer cathode surface exposed, a secondary lithium containing source (10) is brought into close proximity and ionic contact via a secondary electrolyte (8) with the primary cathode (6). The initial formation process then proceeds by driving lithium ions from the secondary lithium source (10), through the secondary electrolyte (8), primary cathode material (6), across the cell electrolyte (4), and into the carbon anode (2), in order to obtain a partial charging and substantial formation of the SEI layer between the cell electrolyte (4) and carbon anode (2). Once the partial charge is complete, the secondary source of lithium (10) and electrolyte (8) are removed and the traditional aluminum foil current-collector is placed in contact with the outer cathode surface as usual for final cell assembly. After final packaging and sealing the completed cell can then continue with further charging and testing as required.
Development of this innovative pre-charging technique has enabled Electrovaya to shift their focus away from the lithium loss issue more toward the development of substantial SEI layer formation techniques that can be more robust than other manufacturers predominantly occupied by lithium loss issues. Formation of a sound SEI layer is crucial for reliable enduring battery function providing a barrier of separation between the electrolyte and carbon electrode. Once formed, the SEI resists any further interaction between these two phases while facilitating the transport of lithium ions between the carbon electrode which carries only individual Li atoms and the bulk electrolyte which carries larger lithium ion salt molecules such as Lithium Hexafluorophosphate (LiPF6)
Elimination of the inactive cathode-material-content and restrictions on the amount of lithium and electrolyte consumed during formation of the SEI layer presents a significant technological advancement for producing cheaper, smaller, lighter cells with superior SEI layer formations effective for use with all cathode materials commonly used today and new cathode materials in the future.